
This one-to-one ratio of charges is not, however, the most common state for many elements. The overall charge on the atom is zero, because the magnitude of the negative charge is the same as the magnitude of the positive charge. The electrovalent bond between ions results from the electrostatic attraction of opposite charges.\) and hypochlorite is ClO −. This is because the number of electrons (negative in charge) is equal to the number of protons (positive in charge).After fluorine gains the electron from sodium, it becomes a negative ion (9 p + 10 e- = -1).After sodium losses the electron, it becomes a positive ion (11 p + 10 e- = +1).Fluorine gains one electron to possess an octet. Here, Na loses one electron to possess an octet. Determine the numbers of electrons that the atoms will lose and gain by the Octet Rule.Solution: Stepwise formulation of the compound is as follows, Q 1] Determine the formula of a compound formed by the reaction of sodium and fluoride. Solved Examples for Ionic Compound Formula Each ion becomes surrounded by solvent molecules.Įlemental atoms generally lose, gain, or share electrons with other atoms so as to attain an equivalent electron structure because of the nearest rare gas with eight electrons within the outer level. For an ionic compound to dissolve, the electrostatic bonds holding the ions in their rigid space lattice must get replaced by attractions between ions and solvent molecules. 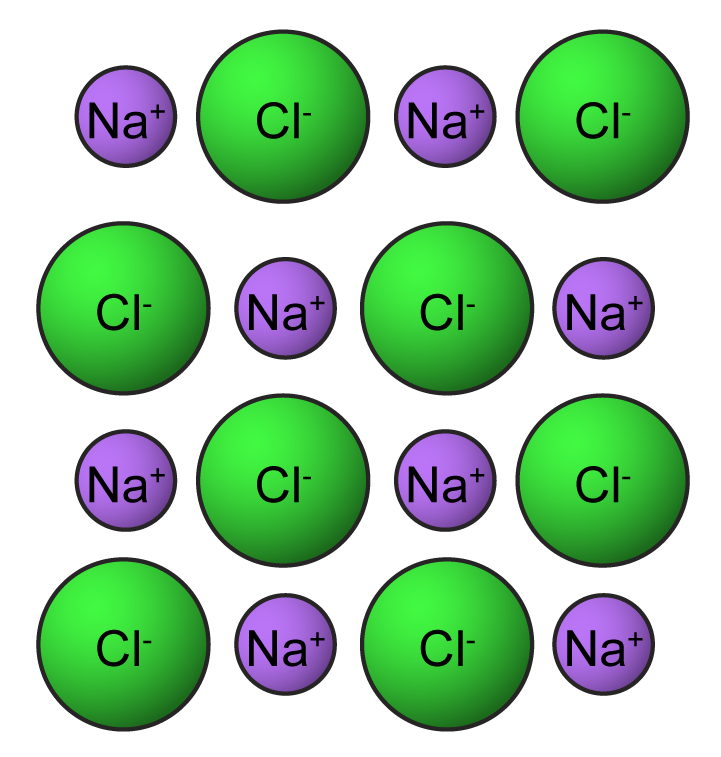
In an ionic compound, like common salt, NaCl, or magnesia MgO, the formula tells us the right ratio of elements present, but it doesn’t specify the unit. Polyatomic ions may be either positive or negative, for example: NH 4 + (ammonium) cation SO 4 2 - (sulfate) anion The principles of ionic bonding with polyatomic ions are the same as those with monatomic ions. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. The group of atoms as a whole acts as a charged species in forming an ionic compound with an oppositely charged ion. Forming an ionic bond, Li and F become Li + and F ions. The sum of the charges of the positive ions (cations) must always equal to sum of the. There’s always a minimum of a little degree of covalent bonding character in ionic compounds. Electron transfer between lithium (Li) and fluorine (F). Ionic compounds always contain a positive ion and a negative ion.2. However, absolutely pure ionic bonding doesn’t exist. This is often different from covalent bonding, during which electrons are shared by atoms, forming directional bonds. It is often an easy positive-negative Coulombic attraction between point charges. Source: Ionic bonding isn’t directional in its purest form. If the ionic compound is soluble during a solvent, we get as a solid compound by evaporation of solid solvent from this electrolyte solution. And, therefore, the process are often thermodynamically understood using the Born–Haber cycle. In some reactions between highly reactive metals (usually from Group 1 or Group 2) and highly electronegative halogen gases, or water, the atoms are often ionized by electron transfer. Ionic compounds without these ions also are referred to as salts and may be formed by acid-base reactions. And people containing basic ions hydroxide (OH−) or oxide (O2−) are bases. Ionic compounds containing hydrogen ions (H+) are acids. It also can include the electron transfer reaction of reactive metals with reactive non-metals, like halogen gases.  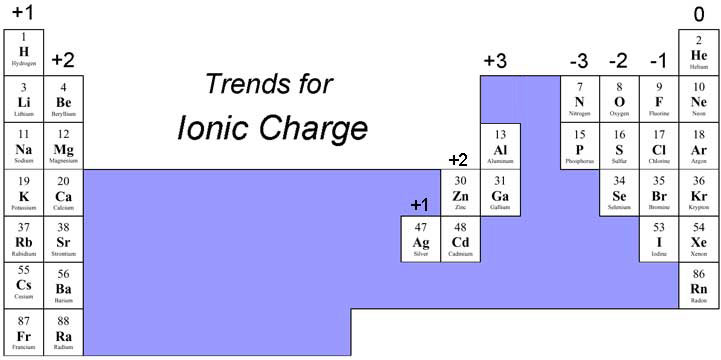
Production of ionic compounds is often from their constituent ions with the assistance of evaporation of their solvent, precipitation, freezing, a solid-state reaction. Ionic compounds are insoluble or have very low solubility in non-polar solvents. they’re nearly always electrically insulating as they’re solids. Ionic compounds have high melting and boiling points.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
